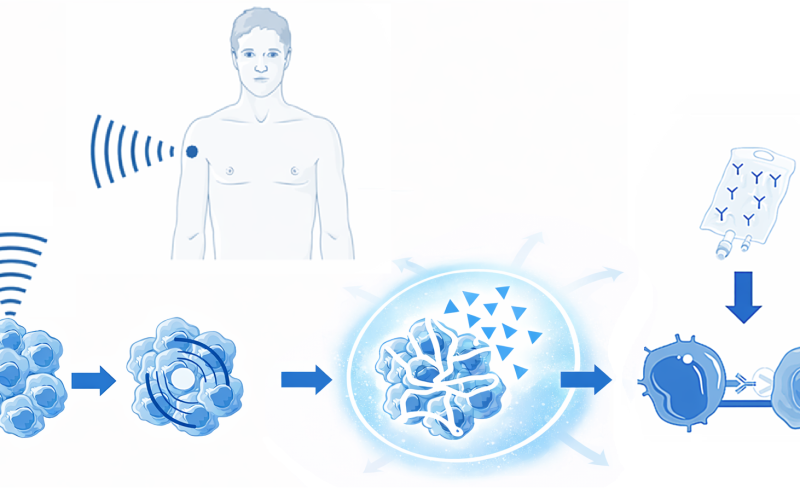
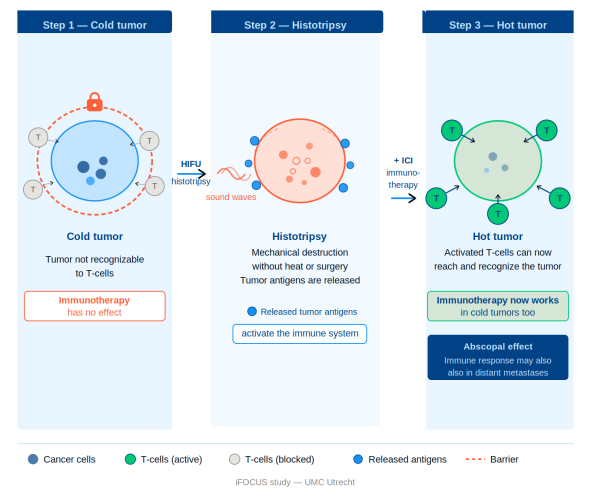
No. The iFOCUS study is an early-phase clinical trial (phase 1). The primary goal is to determine whether the treatment is safe and feasible. Whether and when this combination treatment may become routinely available cannot be determined at this time. The second phase of the study, in which eighteen additional patients will be treated, is currently ongoing. Enrollment is still open for patients who have no other adequate treatment options and who have a tumor or metastasis located less than 3 cm beneath the skin. Physicians are welcome to contact the research team.